Charged particles exert forces on each other. The electric field E = F/q produced by a charged particle at some position r in space is a measure of the force F the particle exerts on a test charge q, if we place the test charge at r. The electric field E is a vector. The electric field due to a charge distribution is the vector sum of the fields produced by the charges making up the distribution. When we think about electricity in everyday life, we seldom think about the electric fields. We are more familiar with the concepts of voltage, current, and power.
When a particle with charge q is placed in an external electric field E, (i.e. an electric field produced by other charges), then an electric force F = qE will act on it. If this force is not balanced by other forces, then the particle will accelerate, and its kinetic energy will change. The electric force will do positive work on the particle. If the electric force is balanced by another external force Fext= -qE, and this external force moves the particle against the electric force, than the external force Fext does positive work.
Recall!
Therefore the work done done by an external force balancing the electric force is
W = ∫Fext·dr= -q∫E·dr.
The work done by the external force is equal to the change in the electrostatic potential energy of the particle in the external field. The change in the potential energy of a charge q when being moved from point A to point B, is the work done by an external force in moving the charge.
∆U = UB - UA = -q ∫ABE·dr.
The integral is taken along a particular path. But the electrostatic force is a conservative force. The integral is independent of the path. ΔU therefore depends only on the endpoints A and B of the path, not on the actual path itself.

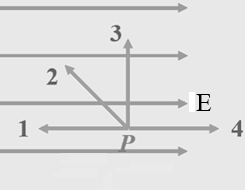
(a) Which requires zero work, to move a positive point charge from
point P to point 1, 2, 3, or 4? All those points are the same
distance from P.
(b) Which requires the most positive work done by an external force, to
move a positive point charge from point P to point 1, 2, 3, or 4?
Discuss this with your fellow students in the discussion forum!
Review the concept of work in physics.
Discuss the relationship between the work done by and against the electric field
and the electrostatic potential energy.
The change in potential energy is proportional to the charge q. Its sign depends on whether the charge is positive or negative. We define the potential difference or voltage ∆V as the potential energy difference divided by the charge, or the potential energy difference per unit (positive) charge. As in the case of gravity, the zero of the potential energy and therefore the zero of the potential are not uniquely defined. Only potential energy differences and potential differences are unique. The SI unit of energy is J, therefore the SI unit of potential is J/C. We define Volt as V = J/C.
The electrostatic potential is a scalar, not a vector.
We say that a charge distribution, which produces an electric field, also produces an electric potential. The electrostatic potential produced by a finite charge distribution is, by convention, set to zero at infinity. Then the potential V(r) of the distribution is the work done per unit charge in bringing a small test charge from infinity to position r. For a charge distributions which extend to infinity, we cannot set the zero of the potential at infinity, because then the potential would be infinite everywhere, and it would be a useless concept. We then set the zero at some convenient reference point, but we always must specify the reference point along with the potential, since there is no unique convention. For many applications we choose the ground to be the zero of the potential.
 The potential difference between the two terminals of an A, B, C, or D cell
battery is 1.5 V. For every Coulomb of negative charge that is moved from
the positive to the negative terminal, 1.5 J of work must be done against
electric forces, and 1.5 J of some other form of energy is converted into
electrostatic energy. When the terminals are connected by a wire, then the
charge is free to move inside the wire, and the electric field does work on the
charge. This work can now be reconverted into some other form of energy.
The potential difference between the two terminals of an A, B, C, or D cell
battery is 1.5 V. For every Coulomb of negative charge that is moved from
the positive to the negative terminal, 1.5 J of work must be done against
electric forces, and 1.5 J of some other form of energy is converted into
electrostatic energy. When the terminals are connected by a wire, then the
charge is free to move inside the wire, and the electric field does work on the
charge. This work can now be reconverted into some other form of energy.
The potential at a point r of a positive point charge located at the origin is the work that must be done per unit charge in bringing a test charge from infinity to r.
V(r) = -∫∞r E·dr = ∫r∞E·dr.
We can bring the test charge along an arbitrary path, which we can think of as being made up of infinitesimal small steps, either in the radial direction or perpendicular to the radial direction. We do not have to do work when we step perpendicular to the radial direction, because then E is perpendicular to dr. Along the radial direction E·dr = Edr, because E and r points both point outward. The integral therefore becomes
V(r) = ∫r∞Edr = ∫r∞(keq/r2)dr = keq/r = q/(4πε0r).
The potential due to a point charge is proportional to the inverse of the distance from the point charge.
If the point charge is not located at the origin, but at position r', we write
V(r) = kq/|r - r'| = kq/((x - x'2) + (y - y'2) + (z - z'2))1/2
The potential outside a spherically symmetric charge distribution with total charge q is the same as that of a point charge q. To find the potential due to a collection of charges, we use the principle of superposition and add the potentials due to the individual charges. Because the potential is a scalar, and not a vector, we just have to add numbers.
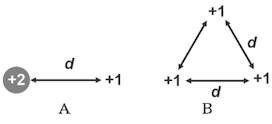
The charges in the groups A and B below are all given in units of Q. Which group of charges took less work by an external force to bring together from infinity?
Solution:
1 electron volt (eV) = 1.6*10-19 J.
1 eV is the change in potential energy of a particle with charge qe = 1.6*10-9
C when the change in potential is 1 Volt (V). The unit was defined so that
when you know the voltage between two points in space, you know the change in
potential energy of an elementary particle when it moves from one to the other
point. (The sign of the change in potential energy depends on the sign of the
charge.) When a free proton moves through a potential difference of 1 V
its kinetic energy decreases by -qV = (1.6*10-19 C)*(1 J/C) = -1.6*10-19 J.
When a free electron moves through the same potential difference of 1 V its
kinetic energy increases by -qV = -(-1.6*10-19 C)*(1 J/C) = 1.6*10-19 J.
Just as we described the electric field around a charged object by field lines, we can also describe the electric potential pictorially with equipotential surfaces (contour plots). Each surface corresponds to a different fixed value of the potential. Equipotential lines are lines connecting points of the same potential. They often appear on field line diagrams.
Equipotential lines are always perpendicular to field lines.
Equipotential lines are therefore perpendicular to the force experienced by a charge in the field. If a charge moves along an equipotential line, no work is done. If a charge moves between equipotential lines, work is done.
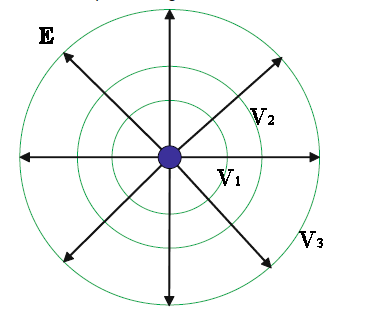 Example:
Example:Field lines and equipotential lines for a positive point charge are shown in the figure. This is a 2-dimensional representation, a cut through the 3-d surfaces.
Please also explore this 3-dimensional representation below. Please click on the image!
In the Bohr model of the hydrogen atom, the electron orbits the proton at a distance of r = 5.29*10-11 m. The proton has charge +qe and the electron has charge -qe, where qe = 1.6*10-19 C. How much work must be done to completely separate the electron and the proton
An alpha particle containing two protons is shot directly towards a platinum nucleus containing 78 protons from a very large distance with a kinetic energy of 1.7*10-12 J. What will be the distance of closest approach?
Solution:
An evacuated tube uses a voltage of 5 kV to accelerate electrons from rest to
hit a phosphor screen.
(a) How much kinetic energy does each electron gain?
(b) What is the speed of an electron when it hits the copper plate?
Solution:
The electrostatic potential V is related to the electrostatic field E. If the electric field E is known, the electrostatic potential V can be obtained using V = -∫E·dr.
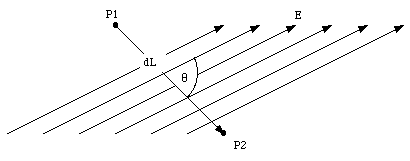
Consider the two points P1 and P2 shown in figure above. Assume that they are separated by an infinitesimal distance dL. The change in the electrostatic potential between P1 and P2 is given by dV = -E∙dL = -E dL cosθ. Here θ is the angle between the direction of the electric field and the direction of the displacement vector dL. We can rewrite this equation as
dV/dL = -E cosθ = -EL.
EL indicates the component of the electric field along the
direction of dL. If the direction of the displacement is chosen to
coincide with the x-axis, this becomes
dV/dx = -Ex.
For the displacements along the y-axis and z-axis we obtain dV/dy = -Ey
and dV/dz = -Ez.
The total electric field
E can be obtained from the electrostatic potential V
by combining these three equations.
We say that E is the
negative gradient
of the potential V.
E = -(i∂V/∂x + j∂V/∂y + k∂V/∂y) = -∇V.
Note: The derivatives above are partial derivatives. When taking the derivative of V with respect to x, we assume that y and z are constant, when taking the derivative of V with respect to y we assume that x and z are constant, and when taking the derivative of V with respect to z we assume that x and y are constant.
In many electrostatic problems the electric field due to a certain charge distribution must be evaluated. The calculation of the electric field can be carried out using two different methods:
In many cases the second method is simpler, because the calculation of the electrostatic potential involves an algebraic sum, while the direct calculation of the field involves a vector sum.
In some region of space, the electrostatic potential is V(x,y,z) = x2 + 2xy. Find E(x,y,z).
Solution:
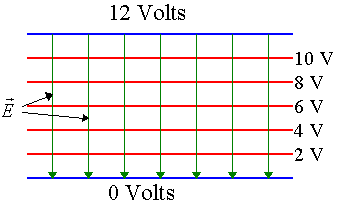 Problem:
Problem:The potential difference between the two plates of the capacitor shown below is 12 V. Equipotential surfaces are shown. If the separation between the plates is 1 mm, what is the strength of the electric field between the plates?
Solution:
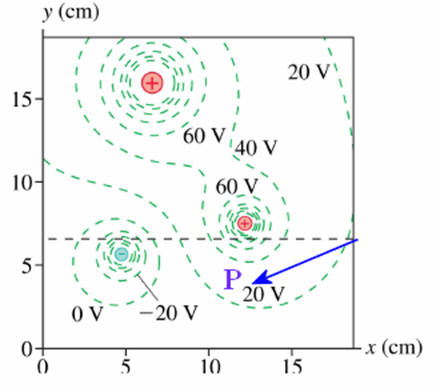 The
graph on the right shows a contour map of the equipotential surfaces due to 3
point charges. Estimate the magnitude and direction of the electric field
at point P.
The
graph on the right shows a contour map of the equipotential surfaces due to 3
point charges. Estimate the magnitude and direction of the electric field
at point P.
Solution: