 Often the system from which we
want to extract heat to do work is a gas. When a gas expands, how
much work does it do on its surroundings?
Often the system from which we
want to extract heat to do work is a gas. When a gas expands, how
much work does it do on its surroundings? Often the system from which we
want to extract heat to do work is a gas. When a gas expands, how
much work does it do on its surroundings?
Often the system from which we
want to extract heat to do work is a gas. When a gas expands, how
much work does it do on its surroundings?
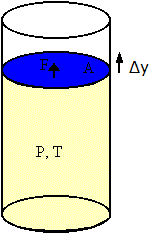
Consider an ideal gas in a cylinder with a movable piston. The gas occupies a volume V, the temperature is T, and the pressure is P. Assume that the piston has a cross-sectional area A. Assume the gas expands by a small amount and the piston moves out by a small amount Δy. The gas exerts a force F = PA on the piston and therefore does work ΔW = FΔy on the piston.
ΔW = FΔy = PAΔy = PΔV
ΔV is the change in volume of the gas. ΔV is positive because the gas expands. (If the gas is being compresses, then ΔV is negative and work is done on the gas.)
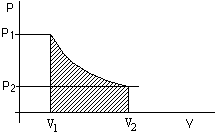 If the volume of the gas changes from V1 to V2,
then the total work done by the gas depends on how exactly the pressure varies during the
expansion process.
If the volume of the gas changes from V1 to V2,
then the total work done by the gas depends on how exactly the pressure varies during the
expansion process.
W = ∑jPjΔVj. We sum the
work done during a large number of small changes of the volume.
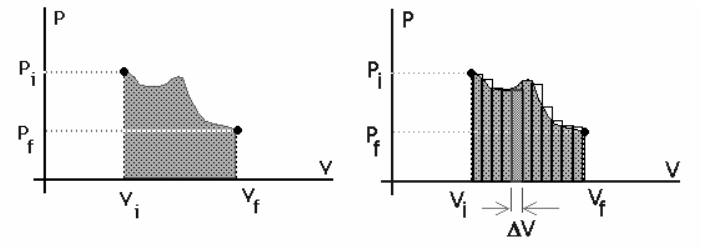
If the pressure and volume are known at each step of the expansion process, then the work can be represented as the area under the curve on a PV diagram.
AI Study Tip:
Example prompt: 'I have a problem where an ideal gas expands isobarically. It does 300 J of work and absorbs 800 J of heat. Walk me through the steps to find the change in internal energy and explain what this means for the temperature of the gas.'
 Problem:
Problem:Solution:
 Problem:
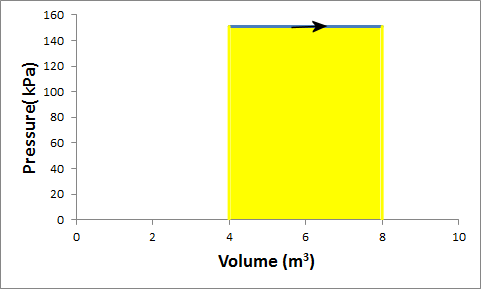
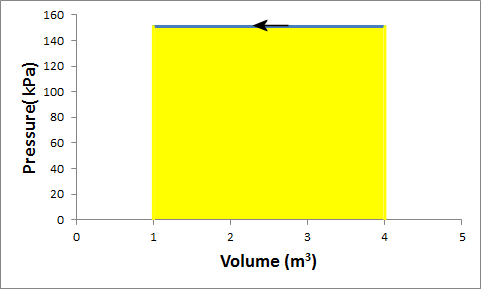
Problem:G in a container is at a pressure of 1.5 atm and a volume of 4 m3.
What is the work done by the gas if it is compressed at constant pressure to 1/4 of its initial volume?
Solution:
 Problem:
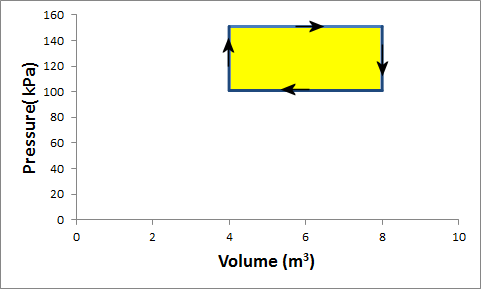
Problem:Gas in a container is at a pressure of 1.5 atm and a volume of 4 m3. What is the net work done by the gas if it goes through the following cycle?
(i) The gas expands at constant pressure of 1.5 atm to a volume
of 4 m3.
(ii) At constant volume of 4 m3 the pressure is reduced
to 1 atm.
(iii) The gas is compressed at constant pressure of 1 atm to
a volume of 4 m3.
(iv) At constant volume of 4 m3 the pressure is
increased to 1.5 atm.
Solution:
The net work done by the gas in one cycle of a process that returns the gas to its initial condition is the area inside the closed loop in the PV diagram. If the loop is traversed in a clockwise direction the net work is positive. If the loop is traversed in a counterclockwise direction the net work is negative.
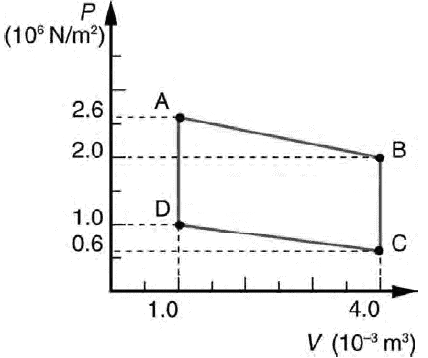
Calculate the net work output of a heat engine following path ABCDA in the
figure on the right.
Solution:
A gas is compressed at a constant pressure of 0.8 atm from 9 liter to 2 liter.
(1 liter = 10-3
m3) In the process, 400 J of thermal energy leaves the gas.
(a) What is the work done by the gas?
(b) What is the change in its internal energy?
Solution:
One mole of an ideal gas does 3000 J of work on its surroundings as it
expands isothermally to a final pressure of 1 atm and volume of 25 L.
Determine
(a) the initial volume and
(b) the temperature of the
gas.
Solution:
AI Study Tip:
Example prompt: 'Explain the difference between an isothermal, adiabatic, and isochoric process. For each, tell me ΔQ, ΔW, or ΔU is zero and provide a real-world example of each.'
A bicycle ergometer is an apparatus that
resembles a bicycle and is used to measure the amount of work done by a person
while pedaling to rotate a large flywheel, usually the front wheel of a
stationary bicycle, against a frictional force. The work done to overcome the
frictional force is converted into thermal energy, which causes the temperature
of the flywheel and of other components in contact with the system to increase.
A nylon belt, which is under tension and wound around the wheel, can provide the
frictional force. The tension can be measured with a calibrated spring. The
work W done by the person on the ergometer is the product of the force due to
the tension F in the belt times the distance through which this force acts. The
force is tangential to the rim of the wheel. The distance is therefore 2π times
the radius of the flywheel r times the number of revolution N made by the
wheel. We have W = NF2πr. For the ergometer we have from the first law of
thermodynamics ΔU = ΔQ - ΔW. ΔW is positive if the system does work on its
surroundings and is negative if work is done on the system. Here ΔW is
negative, since the person does work on the ergometer. ΔW = -W. ΔQ is the
heat put into or taken out of the system. We have ΔU - ΔQ = NF2πr. If we
neglect the heat lost by the system to its surroundings and set ΔQ = 0, then the
increase in the thermal energy of the system equals ΔU = NF2πr. The temperature
of the flywheel therefore increases by ΔT = ΔU/mc, where m is the mass of the
flywheel and c is its specific heat capacity.
The ergometer converts useful work into thermal energy. By analyzing the air
exhaled by the person pedaling the ergometer, the rate at which the body uses
chemical energy can be measured. By knowing how much chemical energy the body
uses at rest and how much it uses while pedaling, the efficiency of the body in
converting chemical energy into useful work can be determined.
There is no change in the internal energy of an ideal gas undergoing an isothermal process since the internal energy depends only on the temperature. Is it therefore correct to say that an isothermal process is the same as an adiabatic process for an ideal gas? Explain your answer.
Discuss this with your fellow students in the discussion forum!